Certificates and Registrations
Novodiax continues to rapidly develop and deliver safe, effective and reliable products for In Vitro Diagnostic (IVD) use! Please see our relevant certificates and registrations below for US and international distribution.
Contact Us
Certificate of IVD / cGMP manufacturing
Our IVD Test Menu Continues to Grow:
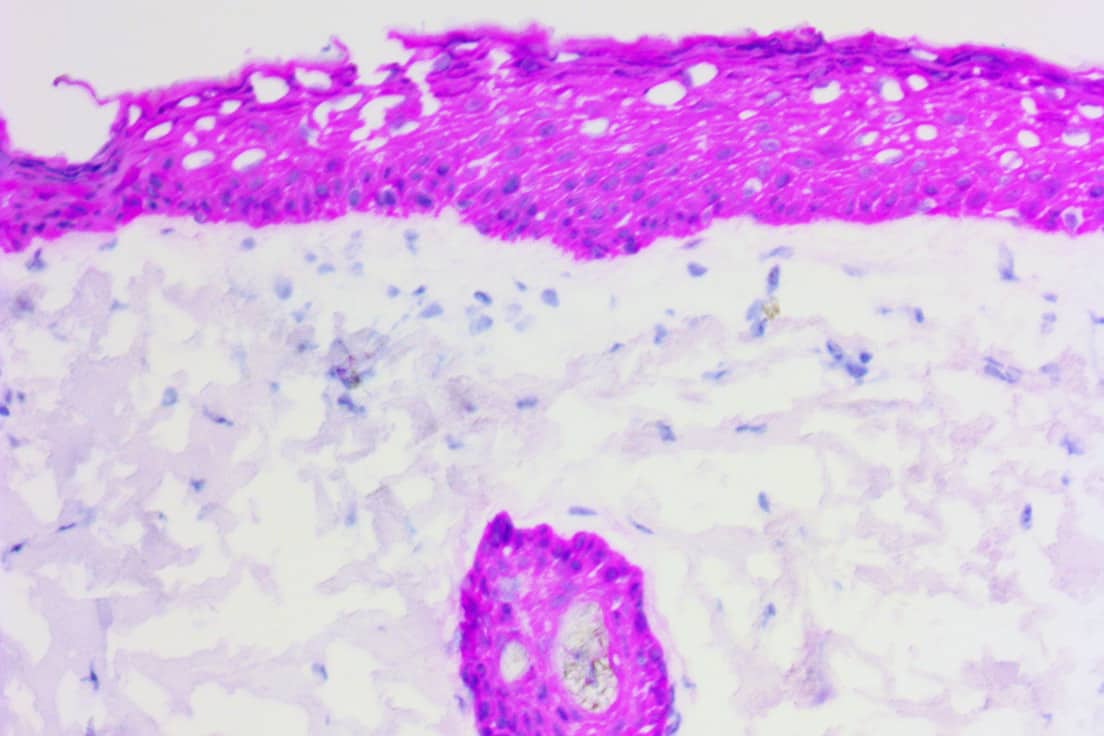
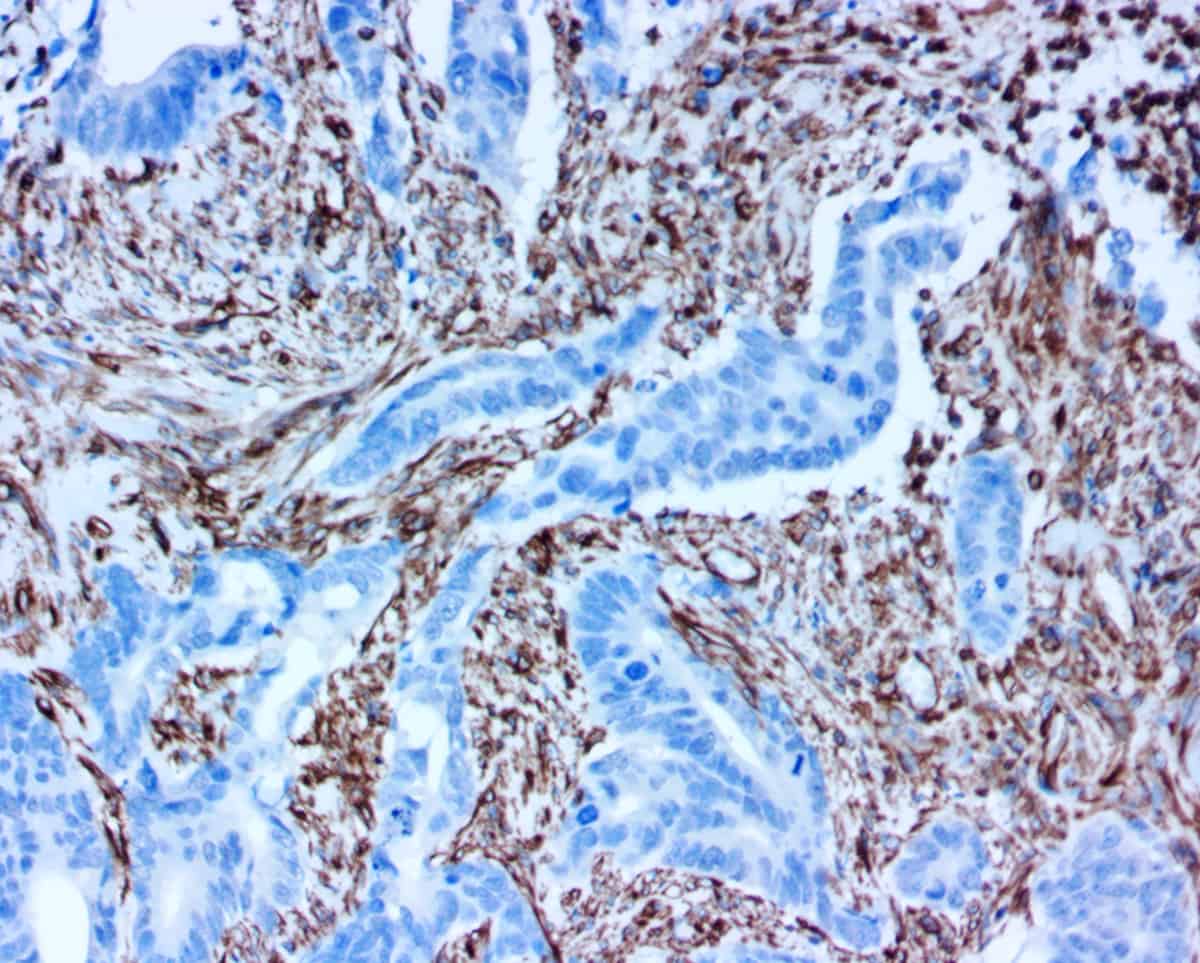
Novodiax ihcDirect® family of products FDA Listed:
AE1/AE3, Calponin, CEA, CD20, CD45, CK5, CK7, CK 8/18, CK19, CK20, Pan-CK 4Abs, EMA, GFAP, Ki67, Mart-1, Podoplanin, SMMHC, and Vimentin.
Read more…
Our Continued Focus Upon Quality:
Novodiax continues to adapt and improve our Quality Systems to cover a wider range of products including instrument systems.
Read more…
California Department of Public Health: Food and Drug Branch
Read more…